If the adhesive forces between water molecules and the molecules of the surface are weak compared to the cohesive forces between the water molecules, the water does not “wet” the surface. Consider what happens when water comes into contact with some surface. The IMFs of attraction between two different molecules are called adhesive forces.

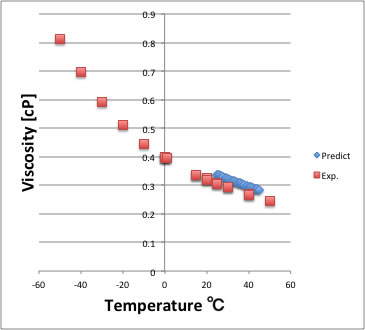
Some insects, even though they are denser than water, move on its surface because they are supported by the surface tension. A steel needle carefully placed on water will float. As a result of this high surface tension, the surface of water represents a relatively “tough skin” that can withstand considerable force without breaking. Among common liquids, water exhibits a distinctly high surface tension due to strong hydrogen bonding between its molecules. This property results from the cohesive forces between molecules at the surface of a liquid, and it causes the surface of a liquid to behave like a stretched rubber membrane. Surface tension is defined as the energy required to increase the surface area of a liquid or the force required to increase the length of a liquid surface by a given amount. Larger drops are more greatly affected by gravity, air resistance, surface interactions, and so on, and as a result, are less spherical. This is because the ratio of surface area to volume is at a minimum in a sphere. A small drop of liquid tends to assume a spherical shape. Because of the unbalanced molecular attractions on the surface molecules, liquids contract to form a shape that minimizes the number of molecules on the surface-that is, the shape with the minimum surface area. However, the molecules on the surface of a liquid are attracted only by about one-half as many molecules. The molecules within a liquid are surrounded by other molecules and are attracted equally in all directions by the cohesive forces within the liquid. The various IMFs between identical molecules of a substance are examples of cohesive forces. Increased thermal energy weakens intermolecular forces, corresponding to molecules moving more freely and liquids thus flowing faster at higher temperatures. Lastly, viscosity depends on temperature. Hydrocarbons of increasing molar masses and lengths interact over greater areas and entangle more frequently, resulting in stronger dispersion forces and higher viscosities.

Viscosity is influenced by molecular shapes. This results in a stronger attraction between glycerol molecules, making glycerol more viscous than methanol. Although both form hydrogen bonds, glycerol has three –OH groups instead of one and forms more hydrogen bonds per molecule. Viscosity expresses a liquid's resistance to flow and is often measured in poise, or grams per centimeter-second,Ĭonsider methanol and glycerol. For mercury, the cohesive forces outweigh the adhesive forces, resulting in a convex meniscus. This phenomenon is called capillary action.įor water, the adhesive forces between water and glass are stronger than the cohesive forces, resulting in a concave, or inward-curving, meniscus. The liquid in the capillary rises until gravity overcomes the adhesive and cohesive forces. Upon placing a narrow capillary tube in water, the water molecules spread along the tube's surfaces via adhesion, increasing the liquid's surface area and drawing the rest of the liquid with it by cohesion. The energy needed to increase a liquid’s surface area by a unit amount is called its surface tension, which is often measured in joules per meter-squared.Īdhesive forces, in contrast, exist between dissimilar molecules, such as water and glass. The interior molecules are more energetically stable than surface molecules because they experience more cohesion, which lowers their potential energy.įluids, therefore, try to reduce their potential energy by minimizing the surface area, creating a surface under tension that behaves like an elastic membrane. In water, the cohesive forces pull interior molecules equally in all directions, resulting in zero net force on average, while surface molecules experience only a downward pull, which causes the molecules to pack closely together.
The attractive forces between similar molecules in a substance are called cohesive forces. Most properties of liquids are dictated by intermolecular forces. Ever wondered why a yen coin, when carefully placed on water, floats?
